Background
The term “Mild Cognitive Impairment: MCI” is used to describe older people with demonstrable cognitive impairment who have not crossed the threshold for dementia (Lopez, 2013). It is characterized by a minor but observable decline in cognitive function and is considered a symptomatic stage before dementia (Zhang et al., 2016). Among older adults, MCI increases the risk of dementia, especially Alzheimer’s disease. In the US, it is an estimated 12% to 18% of people age 60 or older have MCI (Alzheimer's Association, 2022). In Thailand, approximately 71.4% of older people have been diagnosed with MCI (Griffiths et al., 2020). Also, dementia in older people will rise from 1.1 million in 2030 to 2.0 million by 2050 (Leethong-In et al., 2019). Dementia significantly impacts older adults, their families, and carers. It also challenges health professionals throughout the pathway of care. The alternative strategy for reducing the prevalence of dementia is to slow down its onset in older adults at risk for progression to dementia.
While there is no exact cure for MCI and dementia nowadays, it is possible to slow down or prevent the progression from MCI to dementia. Evidence shows the potential benefit of cognitive intervention in older adults. The neurobic exercise concept was postulated by Katz and Rubin (1999). It is a form of brain exercise designed to help keep the brain healthy and active. This exercise is distinct from other types of brain exercises. It involves non-routine or unexpected experiences using various combinations of the five physical and emotional senses. Neurobic exercise help improves nerve impulses and the linkage between various brain data. Moreover, this exercise help stimulates neurons to produce neurotrophins that have chemical effects on nerve cell growth, improve nerve fiber branch and reduce nerve cell degeneration. As a result, neurobic exercise can stimulate other parts of the brain, help prevent the decline in memory performance, and maintain a continuing level of memory performance (Katz & Rubin, 1999).
Previous studies have explored the potential benefit of neurobic exercise. Patani (2020) conducted a randomized clinical trial (RCT) among patients with stroke (N = 40). Participants in the neurobic exercise group had significantly improved cognitive function and quality of life four weeks after completing the intervention than the control group. In another randomized controlled trial with older adults undergoing major, noncardiac, non-neurological surgery (N = 268), after receiving the neurobic exercise intervention, patients showed a decreased incidence of delirium than the controls at seven days post-operation (Humeidan et al., 2021). In addition, in a non-randomized two-group posttest design among older adults with comorbidities (N = 60), the result showed that one month after the intervention, participants in the neurobic exercise group had lower depression scores than the control group (Raj et al., 2020). The evidence showed that nurses play a significant role in delivering such programs or activities for seniors with a wide range of cognitive declines, such as MCI and dementia (Tanglakmankhong, 2021).
In Thailand, a few studies have been conducted on neurobic exercise with older persons during hospital admission and in the community. Kanthamalee and Sripankaew (2014) performed a single-group posttest design testing the effects of neurobic exercise with 22 female dementia patients. The results demonstrated memory improvement one month after the neurobic exercise intervention was completed. Nevertheless, this study did not include a control group and was limited to female participants and one cognitive function domain. The effects of a neurobic exercise program were examined by Napatpittayatorn et al. (2019) in 55 older persons with normal to MCI. The findings demonstrated that, at six months after the intervention’s completion, participants in the neurobic exercise group had better cognitive performance and serum Brain-Derived Neurotrophic Factor (BDNF) levels than those in the control group. Kansri et al. (2018) investigated the effects of neurobic exercise on depressive symptoms in 60 older persons with MCI. The finding shows that neurobic exercise programs significantly reduced depression among community-dwelling older persons with MCI. Kriengkaisakda and Chadcham (2012) developed a brain training program based on the neurobic exercise theory to target the short-term memory of patients with mild dementia (N = 34). The result indicated that the brain training program could improve short-term memory among these patients. Wongkhamchai and Pantong (2017) examined the efficiency of the neurobic exercise program in 120 patients with type 2 diabetes mellitus using a non-randomized two-group posttest methodology. The result showed that the experimental groups’ scores on short-term memory were significantly higher after training than the control groups at three months follow-ups.
The results of all studies showed that memory performance in the elderly with MCI could be improved by neurobic exercise. However, rather than older adults with MCI, most previous research focused on healthy aging, the elderly with dementia, and older adults with chronic illnesses (Kanthamalee & Sripankaew, 2014; Kriengkaisakda & Chadcham, 2012; Wongkhamchai & Pantong, 2017). In addition, the activities focused on health education and only used each physical sense to stimulate the brain (Kansri et al., 2018; Kanthamalee & Sripankaew, 2014). Therefore, it could be illustrated that the literature on nursing interventions aimed at improving health outcomes in older adults, especially those with MCI, is limited and has methodological limitations. Another type of brain training, such as traditional brain exercise performed in the senior club affiliated with Saraburi Hospital in Thailand, was used to prevent or slow the progression of dementia among these members. The traditional brain exercise was a practice-based program derived from expert panels as a part of health promotion to prevent or slow the progression of dementia among older adults. Traditional brain exercise focuses on simple brain exercise and social activities. However, traditional brain exercise has certain limitations because the program has not been empirically verified to be effective and enhance the health outcomes of older adults with MCI. Therefore, the randomized, controlled, two-period crossover design was employed to examine the effectiveness of the neurobic exercise intervention among older adults living in the community. Memory performance, both objective and subjective memory, served as this study’s outcomes.
The study was conducted concerning the COVID restriction. In addition, the risk of confounding effects was minimized because all interventions were delivered and measured on the same participants, and every participant received both treatments. The participants serve as their control. This study’s results would benefit not only older adults with MCI but also those who care for them, such as their families, nurses, and other relevant healthcare providers.
The purpose of this study was to determine the effect of a neurobic exercise program on memory performance among older adults with MCI residing in the community. The specific aims of this study were: 1) To compare the mean differences of subjective and objective memory between the experimental and control groups and 2) To compare the mean differences of subjective and objective memory within the experimental and the control group.
Methods
Study Design
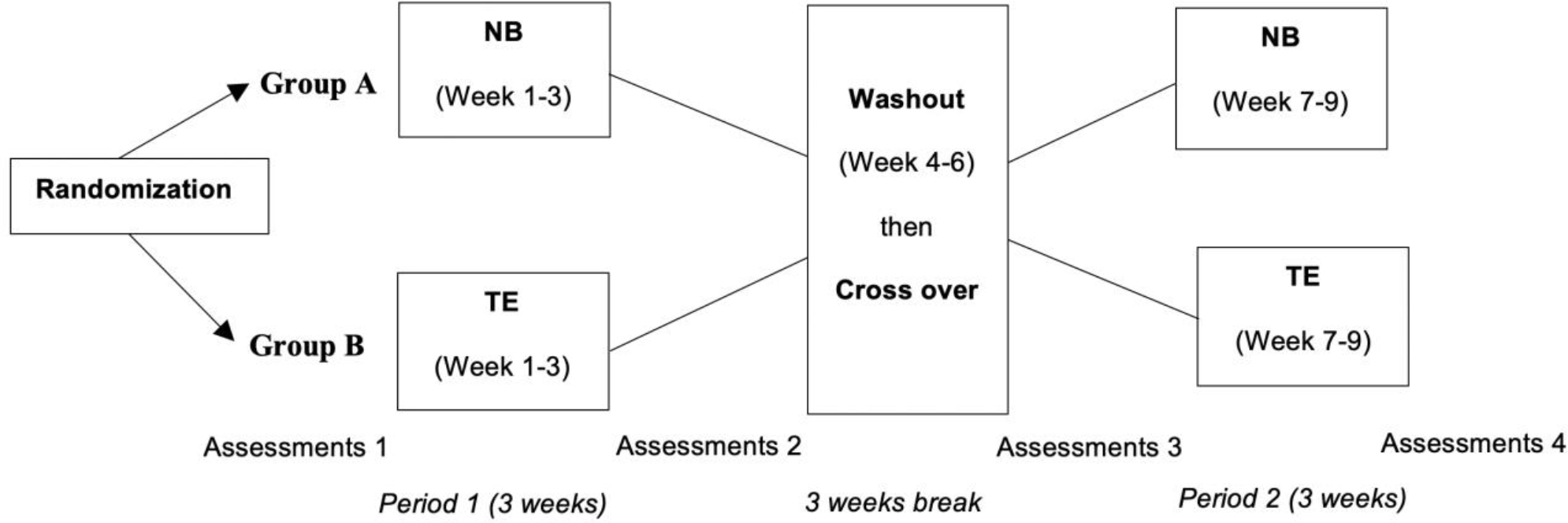
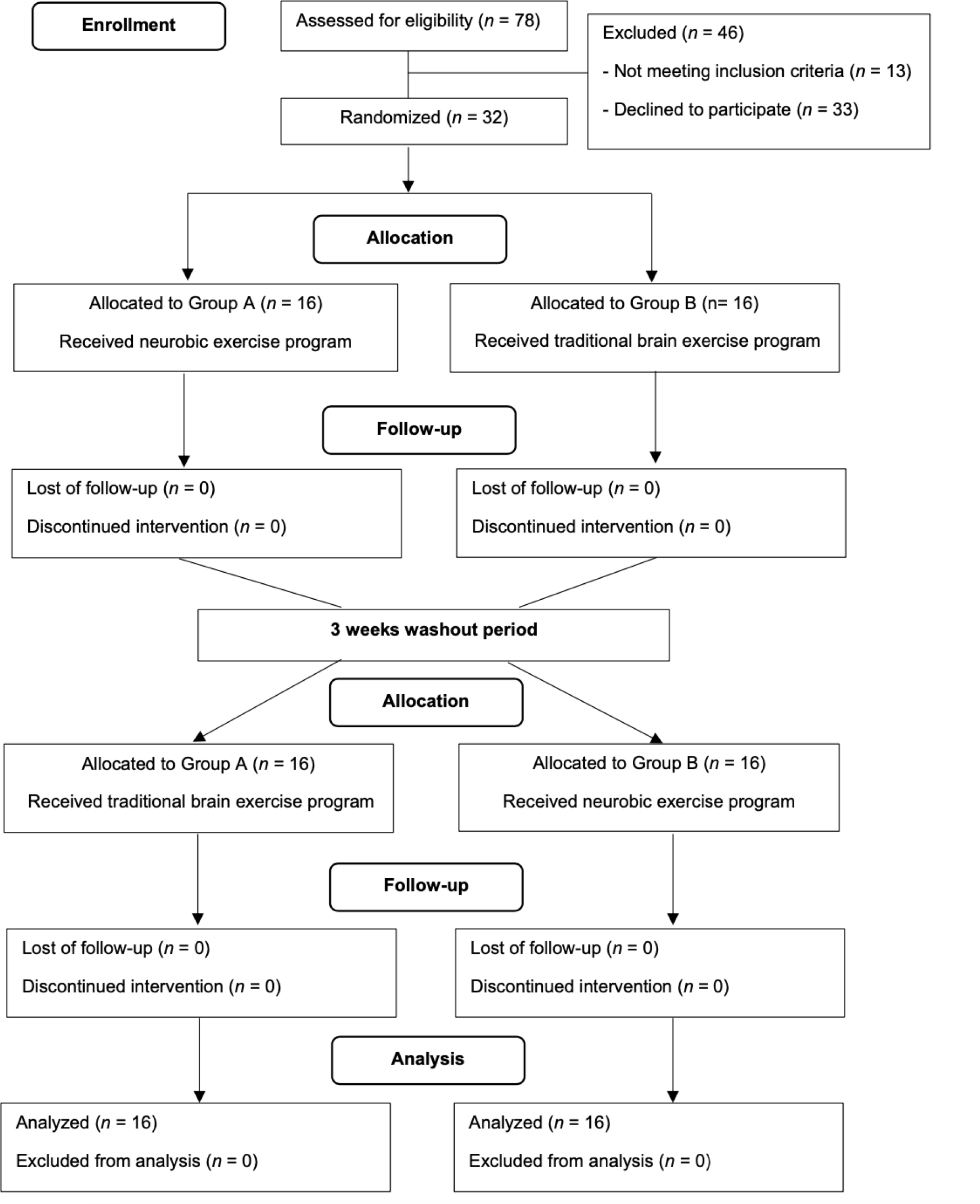
This study employed a randomized controlled trial with a two-period crossover design (two 3-week intervention phases separated by a 3-week washout period). The study was conducted from June 2021 to November 2021. Group A (n = 16) received the neurobic exercise program for three weeks, followed by a three-week washout period, and then the traditional brain exercise program for three weeks. Group B (n = 16) received the treatments in the reverse order but otherwise in a similar manner (Figure 1). This study had three research assistants (RAs), including two research assistants who conducted the screenings and collected the outcome data (RA1-2), and another RA3 was running the traditional brain exercise program. Two Ras (RA1-2) who are registered nurses with at least two years of experience in geriatric care were selected from Saraburi Hospital. The researcher trained two Ras (RA1-2) to collect accurate and consistent data from both groups blindly by not knowing which group was the neurobic exercise program or the traditional brain exercise program. Another RA3, a registered nurse who works in the senior club, was trained to run the traditional brain exercise program. This study was reported in accordance with the Consolidated Standards of Reporting Trials (CONSORT) checklist (Moher et al., 2010) (Figure 2).
Samples/Participants
Older people were recruited from the senior club of Saraburi Hospital in Tambon Pak Phriao, Amphoe Muang of Saraburi province. Among 78 older adults were eligible to participate in this study. Among them, 13 participants did not meet the study inclusion criteria. Of these 78 older adults, 32 were randomly selected and consented to participate in this study. The inclusion criteria consisted of (1) males and females aged 60 years or older, (2) scored <24 on the Montreal Cognitive Assessment (MoCA), (3) scored >12 on the Modified Barthel ADL index (BAI), (4) have a Body Mass Index (BMI) score in the range 18.49-34.9 kg/m2, (5) no history of psychiatric disorders or neurological conditions; no hearing or visual impairment as diagnosed by a physician, (6) ability to write and read as well as communicate well, (7) willingness to participate in the study throughout the program. The exclusion criteria consisted of (1) severe complications or serious neurological or musculoskeletal conditions, (2) serious conditions from comorbidity that made the participant unable to participate in the intervention, and (3) older adults with a history of allergies to the food or flowers used in the activities.
In this study, the G* Power analysis was used to calculate the sample size, with a power of 0.08, α = 0.05, and an effect size of 0.50, according to Napatpittayatorn et al. (2019). The results indicated that the study needed 24 participants. In order to adjust for any participants dropping out or data attrition, an additional 30 percent was added to the sample size for a total of 32 subjects in total. These 32 older adults were randomly assigned into groups A or B, n = 16 for each group, through drawing lots (Figure 2).
Instruments
Three instruments were used in this study. First, the demographic variables include age, gender, marital and living status, education level, occupation, and underlying diseases. Second, the short form of the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE), translated into Thai by Senanarong et al. (2001), was used to measure subjective memory. This test measures the informant’s perception of the older person’s cognitive decline. Older adults are required to compare their performance today with their performance ten years ago. Informants were asked to indicate the change on a 16 items scale from 1 (much improved) to 5 (much worse). The cut-off scores were based on the total score divided by the number of questions (average item score range 1- 5). Lower scores indicated better subjective memory performance (1) A score below 3.00 indicates improvement, (2) 3.00 indicates no change, (3) 3.01 – 3.50 indicates a slight decline; (4) 3.51-4.00 indicates moderate decline; and (5) 4.01 – 5.00 indicates severe decline. The Cronbach’s alpha reliability score of IQCODE for this sample was 0.94. Third, a standardized Common Objects Memory Test (COMT) was used to measure objective memory. The COMT was designed to assess age-related memory impairments in individuals with various educational, language, and cultural backgrounds. The author and a bi-lingual researcher translated the COMT manual instructions into Thai using a translation and back-translation method (Brislin et al., 1973). The translation achieved 100% inter-rater agreement. The COMT (Thai version) was tested with five older adults to ensure familiarity with the common objects and the distracters. The total COMT scores were summed across these three steps, and the possible range was 0-90. Higher scores indicated better objective memory performance. The Cronbach’s alpha of the instrument was 0.80.
Interventions
Neurobic exercise program
The researcher developed the neurobic exercise intervention by integrating the neurobic exercise concept (Katz & Rubin, 1999) and previous evidence-based research (Kanthamalee & Sripankaew, 2014; Napatpittayatorn et al., 2019). The content, process, and arrangement of the neurobic exercise program were validated by five experts in neurological and geriatric care. Further, the researcher tested the neurobic exercise intervention with 20 older adults for its feasibility and acceptability. The pilot study results showed the neurobic exercise intervention was feasible and acceptable among these older adults. The neurobic exercise program utilized diverse combinations of physical and emotional senses. The neurobic exercise program consisted of six sessions 1) Stimulation of visual sense through a tray game, 2) Stimulation of smell sense through the use of natural aroma, 3) Stimulation of hearing sense through listening and identifying nature sounds, 4) Stimulation of the sense of touch through the writing and guessing game and a puzzle box game, 5) Stimulation of taste sense by tasting fruit, food, and desserts, and 6) Integration of multi-senses stimulation; Application in daily usage. The researcher conducted the intervention twice a week for three consecutive weeks. Each session lasted approximately 60 minutes. The activities were done in a group to stimulate the participants’ brains and help them enjoy spending time with others.
Additionally, five homework assignments were given that involved simple brain stimulation activities to activate the participant’s brains when they were on their own. Specifically, participants were trained to practice various activities beyond the daily routine. For example, changing the directions in going out and back home, or going to specific places such as temples, markets, etc., touching the differently shaped objects in their houses while closing their eyes and creating new cooking menus by listing all ingredients and calculating the cost.
Traditional brain exercise program
The traditional brain exercise program was a practice-based program derived from expert panels as a part of health promotion activities in preventing or slowing the progression of dementia among older people. It was implemented in the senior clubs affiliated with Saraburi Hospital in Thailand. Activities based on scientific evidence were employed to improve the cognitive function of older adults. The activities were conducted in small groups, approximately 3-4 persons per group, and focused on brain stimulation. This program consisted of six 60-minute sessions conducted twice weekly for three weeks. The activity schedule was the same period as the neurobic exercise program. The activities consisted of 1) Psychoeducation, “brain exercise to prevent dementia for the elderly”, 2) Drawing and coloring, 3) Paper folding, 4) Singing a song, 5) Photo hunting, and 6) Calculation games.
Data Collection
Two research assistants (RA1-2) collected data from all older adults with MCI in both groups four times at baseline, post-intervention (week 3), follow-up stage (week 7), and the end of the study (week 9). During the intervention and data collection process, no unintentional harm or program-related negative effects were found among the participants.
Data Analysis
All statistical analyses were conducted using IBM SPSS version 26.0. Descriptive statistics were used to describe the characteristics of the participants. Chi-square, Fisher’s exact test, and independent t-test were used to examine the differences in demographic characteristics between these two groups. Repeated measures ANOVA was used to compare the differences in mean scores of the outcomes between the two groups and within the group across the four measured periods.
Ethical Considerations
Ethical approval was obtained from the Research Ethics Committee of Burapha University (IRB3-011/2564) and the Ethical Committee of Saraburi Hospital (EC002/2564), the date of approval Jan 26, 2021. Each participant willingly signed a consent form after learning about the goal of the study, the processes involved, and their rights to confidentiality and withdrawal autonomy. Permission to use the instruments was granted by the original authors before data collection.
Results
Characteristics of the Participants
The baseline demographic characteristics of the sample are presented in Table 1. There was no statistical significance between groups A and B in demographics at baseline. The mean MoCA score was 21.13 (SD = 2.01, range 16-23), the mean ADL score was 19.91 (SD = 0.29, range 19-20), and the mean BMI was 24.56 (SD= 3.61, range 16.01-34.52). The mean age of the participants was 67.53 years (SD = 4.43, range 60-78), 93.8% (n = 30) were female, 46.9 % (n = 15) were married, and 87.5% (n = 28) lived with family members at home. Of the 32 participants, 87.5% (n = 28) had primary education. 81.3% (n = 26) reported having other chronic illness conditions, including hypertension 68.8% (n = 22), dyslipidemia 46.9% (n = 15), diabetes 21.9% (n = 7), and heart disease 3.1% (n = 1) whereas 43.75% (n = 14) reported more than one condition.
| Characteristics | Group A (n = 16) | Group B (n = 16) | t/ X2/F | p-value | ||
|---|---|---|---|---|---|---|
| n | % | n | % | |||
| Age | ||||||
| Range | 60-74 | 60-78 | -0.671a | 0.507 | ||
| Mean (SD) | 67(3.46) | 68.06 (5.29) | ||||
| Sex | ||||||
| Male | 1 | 6.3 | 1 | 6.3 | 0.000b | 0.999 |
| Female | 15 | 93.8 | 15 | 93.8 | ||
| Marriage status | ||||||
| Single | 3 | 18.8 | 4 | 25.0 | 0.210c | 0.901 |
| Married | 8 | 50.0 | 7 | 43.8 | ||
| Divorced/widowed | 5 | 31.3 | 5 | 31.3 | ||
| Living status | ||||||
| Living alone | 2 | 12.5 | 2 | 12.5 | 0.000b | 0.999 |
| Living with other | 14 | 87.5 | 14 | 87.5 | ||
| Education level | ||||||
| Less than high school | 14 | 87.5 | 14 | 87.5 | 0.000b | 0.999 |
| Completed high school | 2 | 12.5 | 2 | 12.5 | ||
| Occupation | ||||||
| Not working | 10 | 62.5 | 12 | 75.0 | 2.186b | 0.624 |
| Working | 6 | 37.5 | 4 | 25.0 | ||
| Income (Thai Baht/month) | ||||||
| Range | 600-10,000 | 600-10,000 | -0.417a | 0.679 | ||
| Mean (SD) | 1393.75 (2320.47) | 1737.50 (2338.91) | ||||
| Chronic illness | ||||||
| No | 3 | 18.8 | 3 | 18.8 | 0.000b | 0.999 |
| Yes | 13 | 81.3 | 13 | 81.3 | ||
Crossover Analysis
Identifying carryover effects between the two study periods was the primary objective of the analysis. The statistical analysis found no carryover effects for either outcome (IQCODE, p = 0.219 and COMT, p = 0.139). It demonstrated that there were no carryover effects and that the washout phase of three weeks was sufficient. After that, significant differences between the intervention effects of neurobic exercise and traditional brain exercise programs were evaluated.
Subjective memory
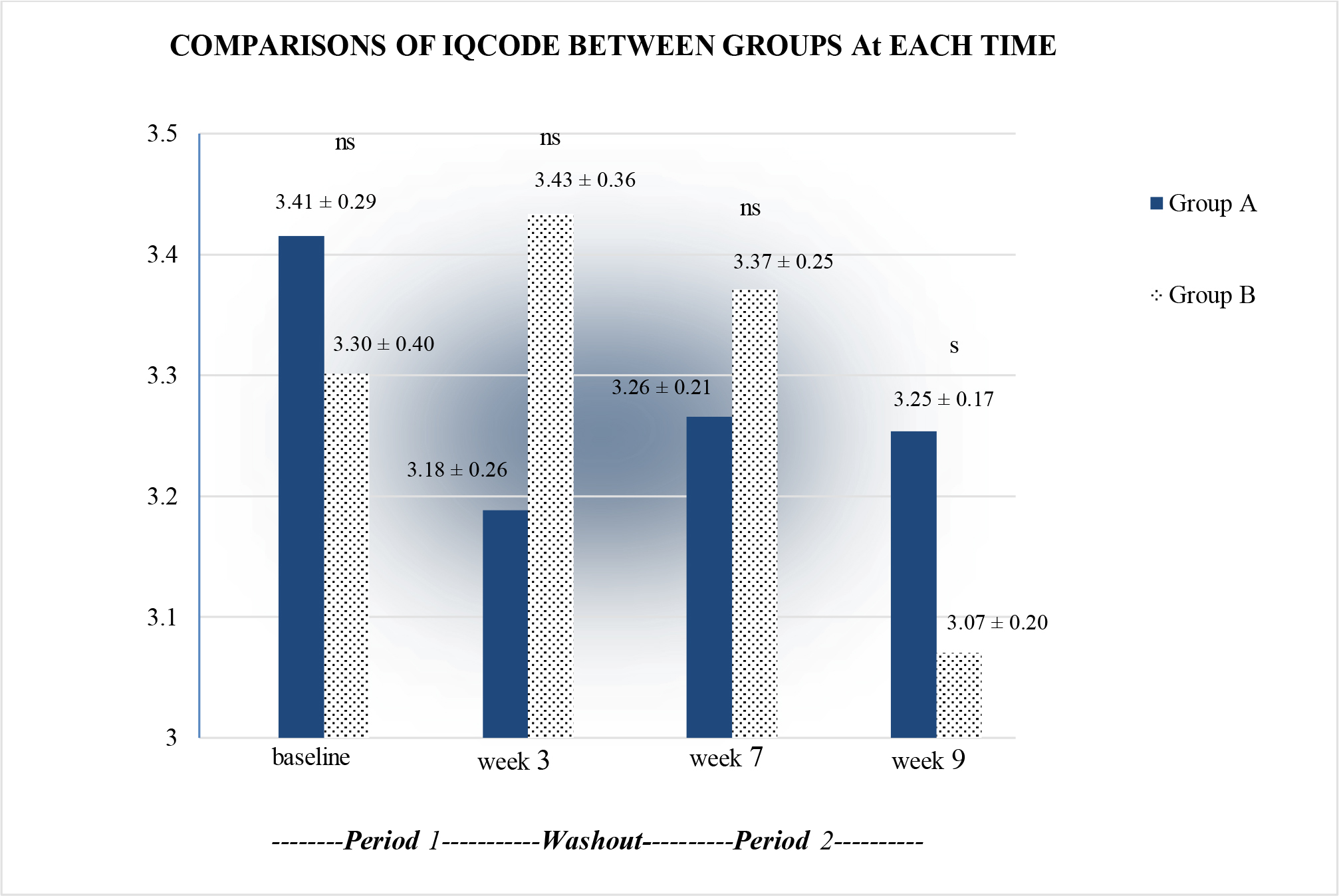
We performed repeated measures ANOVA to demonstrate differences between both groups. For subjective memory (IQCODE), rmANOVA revealed a significant difference of within-subject (F1.437, 43.113 = 9.324, p <0.05) and interaction effect (time*group) (F1.437, 43.113 = 12.313, p <0.05). The comparisons of mean scores measured at different time points found non-significant differences between subjects (F1,30 = 0.023, p >0.05). Then, a post hoc t-test was conducted. For this test, the significance level was adjusted by dividing it by the number of comparisons (0.05/4 (times of comparisons) = 0.0125; Bonferroni correction). Results showed a significant difference in week 9 between the two groups (p <0.0125). Details are presented in Figure 3.
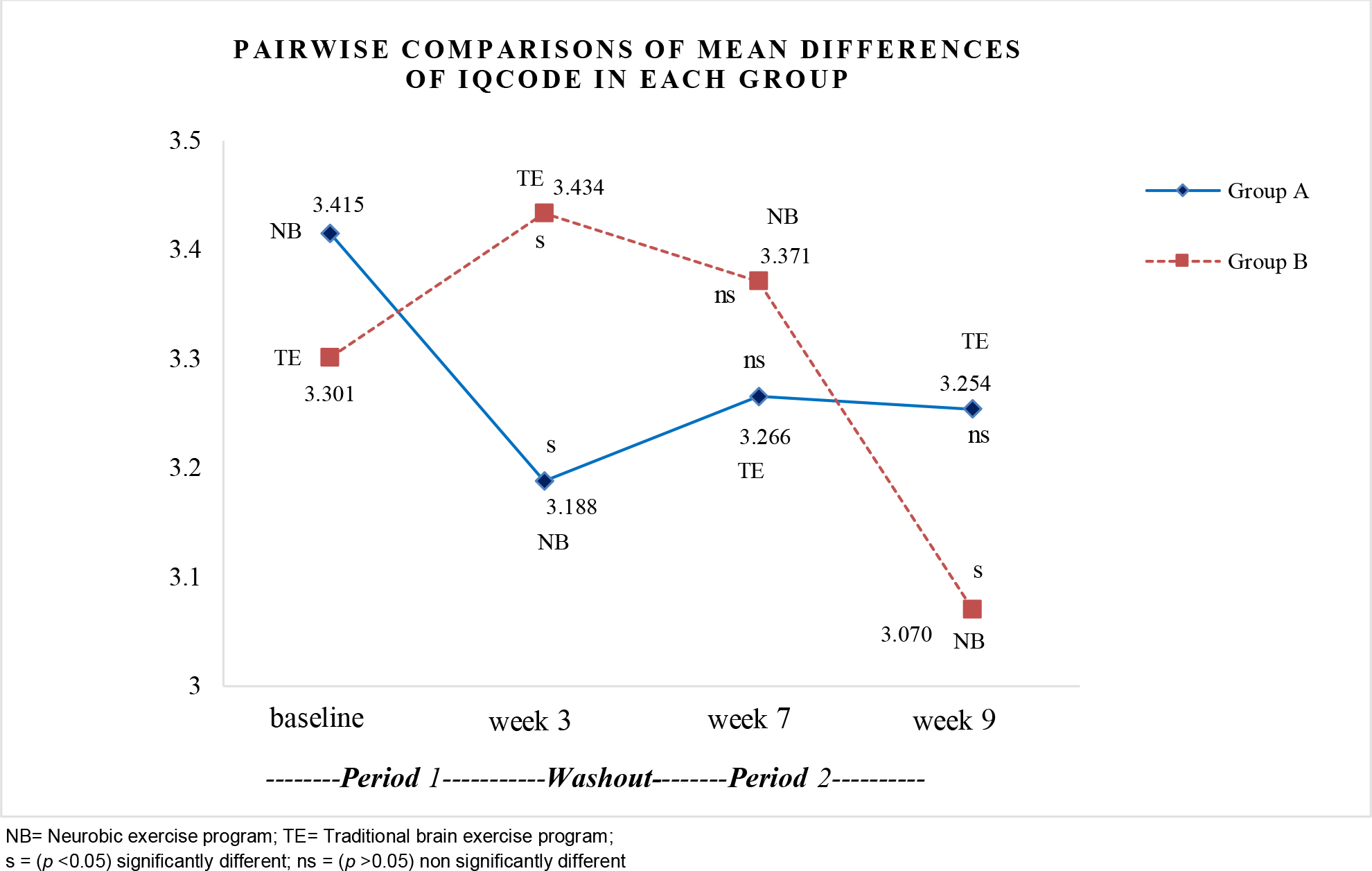
For within the group, when comparing each pair of times for Group A, pairwise comparisons of the mean differences of IQCODE revealed significant differences at baseline and week 3 (p <0.05). After a washout period, there were no significant differences in week 7 and week 9 (p >0.05). In group B, the mean differences of IQCODE revealed significant differences at baseline and week 3 (p <0.05). After a washout period, there were significant differences at week 7 and week 9 (p <0.05). Details are presented in Figure 4.
Objective memory
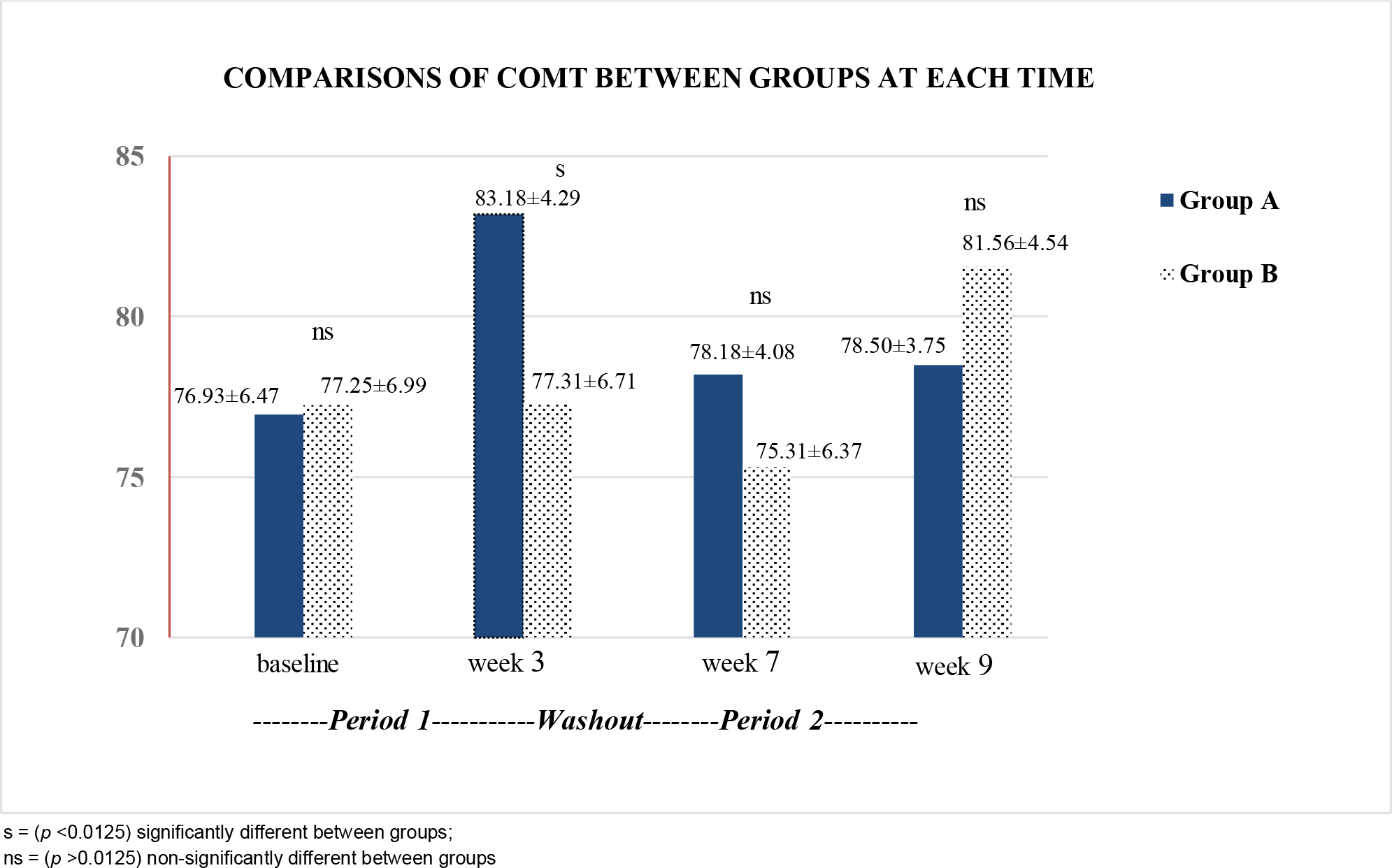
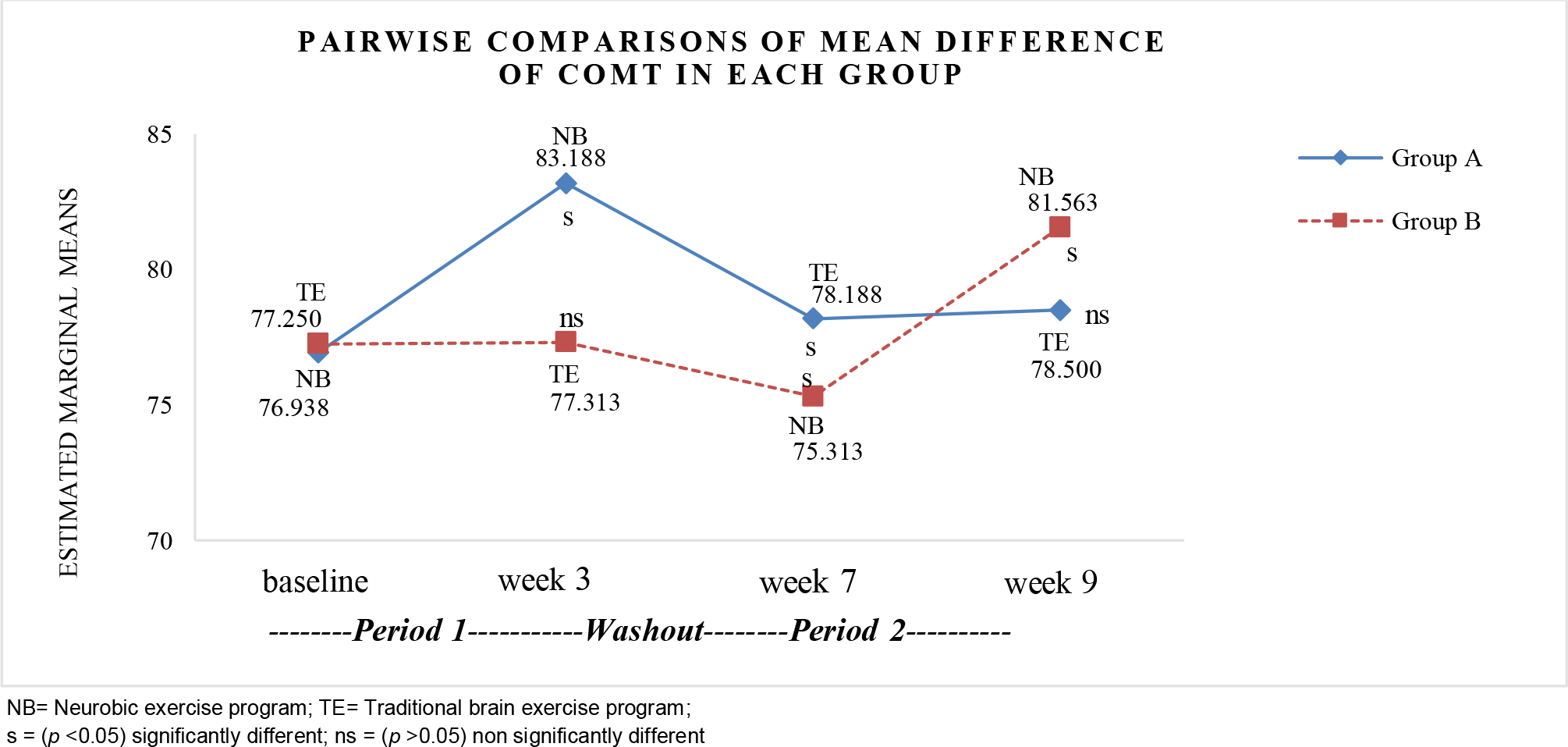
For objective memory (COMT), rmANOVA revealed significant differences of within-subject (F1.794,53.811 = 28.931, p <0.05) and interaction effect (time*group) (F1.794, 53.811 = 31.190, p <0.05). The comparisons of mean scores measured at different time points found no significant difference between subjects (F1,30 = .518, p >0.05). Post hoc t-test showed a significant difference in week 3 between the two groups (p <0.0125). Details are presented in Figure 5.
In group A, when comparing each pair of times, pairwise comparisons of the mean differences of COMT revealed significant differences at baseline and week 3 (p <0.05). However, there were no significant differences in week 7 and week 9 (p >0.05) after a washout period. In group B, the mean differences of COMT showed no significant difference at baseline and week 3 (p >0.05). However, there were significant differences in COMT scores at 7 and week 9 (p <0.05). Details are presented in Figure 6.
Discussion
This study aimed to evaluate the effects of a neurobic exercise program on memory performance in older individuals with MCI. We found that participants who received the neurobic exercise program had lower mean scores of the IQCODE and higher mean scores of the COMT than those in the traditional brain exercise program at post-intervention (week 3) and at the end of the study (week 9). Based on the neurobic exercise concepts proposed by Katz and Rubin (1999) and relevant research, these results could be explained by the fact that the neurobic exercise program was designed to enhance memory performance, both subjective and objective memory, in older adults with MCI. The researcher developed the neurobic exercise program by applying the fundamental components of neurobic exercise to strengthen and enhance the memory performance of the participants. In each session, the fundamental components and techniques of neurobic exercise were utilized to stimulate the brain. If the brain is stimulated by using a variety of physical sense combinations and deviates from a usual activity, the nerve impulses and connections between different neurons in the brain assist the brain in having a steady level of mental fitness, strength, and flexibility (Katz & Rubin, 1999; Scotts, 2013).
Neurobic exercise stimulates neurons to secrete neurotrophins, which have a pharmacological effect on nerve cell proliferation, nerve fiber branching growth, and inhibition of nerve cell degeneration (Napatpittayatorn et al., 2019). In addition, Brain-derived neurotrophic factor (BDNF) is a part of neurotrophic factors. BDNF is found all over the brain, predominantly in the brain areas of the hippocampus, basal forebrain, and prefrontal cortex. These areas are essential for human learning, memory, and other cognitive functions. The BDNF is advantageous for both long-term memory and overall brain function. BDNF helps enhance memory storage and the number, size, and intricacy of dendritic spines (Miranda et al., 2019). BDNF levels are increased by physical activity, maintaining a healthy weight, and leading a mentally and socially engaged, but not unduly stressful, lifestyle. Evidence also demonstrated that neurobic exercise is an alternative method for increasing BDNF levels (Gao et al., 2022).
Moreover, the current study shows that the neurobic exercise intervention can increase BDNF levels in older adults, indicating improved brain function (Napatpittayatorn et al., 2019). According to neurobic exercise, trying to do new things and breaking a routine activity would help strengthen nerve cell stimulation, whereas routine activities use the same brain pathways that could be brain deadening (Napatpittayatorn et al., 2019; Raj et al., 2020). Doing new things or having unexpected experiences help stimulate the production of neurotrophins, which chemically affect the growth of nerve cells and nerve fiber branches, reduce the degeneration of nerve cells, and increase blood flow to the brain. Moreover, emotional sensation stimulates the diencephalon, particularly the hypothalamus, which regulates emotion and encodes memory by laying down memory attention. Emotional sensation also stimulates the limbic system, contributing to memory formation by linking emotional states with memories of physical sensations (Kanthamalee & Sripankaew, 2014; Katz & Rubin, 1999).
These results were consistent with previous research studies that showed neurobic exercise effectively enhanced memory function in the elders. In a study conducted by Kriengkaisakda and Chadcham (2012), patients with mild dementia who completed a neurobic exercise program significantly improved their short-term memory three months after completing the program. One month after completing the neurobic exercise intervention, Kanthamalee and Sripankaew (2014) found an improvement in memory among 22 females with dementia, as measured by the Mini-Mental State Examination (MMSE) score. At a 3-month follow-up, (Wongkhamchai & Pantong, 2017) found that patients with type 2 diabetes mellitus in the neurobic exercise intervention group had significantly enhanced short-term memory scores compared to the control group. According to Napatpittayatorn et al. (2019), the participants in the neurobic exercise intervention group showed significant improvement regarding cognitive function and serum brain-derived neurotrophic factor (BDNF) six months after completing the intervention completion than those in the control group.
Moreover, Patani (2020) discovered that four weeks after completion of the neurobic exercise intervention, patients had significantly better cognitive function and quality of life than the controls. Therefore, previous research supports our findings that a neurobic exercise program can improve both subjective and objective memory in older adults with MCI. However, the results showed that objective memory (COMT) score slightly decreased in the washout period when the participants did not continue the activities to stimulate their brains. Therefore, promoting continuous activities by the older adults themselves to stimulate the brain should be conducted. The evidence showed that if the brain is being used continuously and appropriately stimulated, the brain will improve even with advanced age (Patani, 2020).
This study differs from previous neurobic exercise studies, including activities that stimulate the brain through several senses. The activities presented the brain with non-routine or changed daily activities. These activities can enhance the memory retention of older people and delay the deterioration of the brain. This intervention utilized the five senses and added the emotional sense to each activity. The participants were also encouraged to perform non-routine daily activities. This help promotes continuous growth and strength of the nerve cells. The activities were conducted in small groups and the form of games. Some activities include competitions to encourage teamwork and physical activity among participants. Combining the emotional sense with the five senses will stimulate the hypothalamus, which regulates emotion and encodes memory. It also helps stimulate the limbic system, which contributes to memory formation by integrating the emotional states with stored memories of physical sensations (Kanthamalee & Sripankaew, 2014). Moreover, combining two or more senses could promote the operation of the memory-related regions of the brain, such as the frontal, parietal, temporal, occipital, diencephalon, and limbic systems. As a result, the function of the neural network, which is responsible for both short-term and long-term memory, is enhanced.
At the end of the program, participants in both groups were asked to complete a seven-item Likert-type questionnaire to measure their level of satisfaction with the neurobic exercise and traditional brain training programs. The mean satisfaction scores for the neurobic and traditional brain exercise programs were 34.63 (SD = 0.50) and 33.94 (SD = 1.61), respectively. There were no statistically significant differences in satisfaction between the neurobic and traditional brain exercise programs (p >0.05). In response to open-ended questions, participants reported that all neurobic exercise program activities were appropriate for their age and health conditions. The program activities were beneficial and suited to their needs. Moreover, participants appreciated the neurobic exercise program because it is simple to practice independently and does not require any specialized equipment to activate the brain.
Limitations of the Study
This study was was only conducted at one senior club under the Saraburi Regional Hospital. These older adults at the senior club were more likely to be active and learn new things. In addition, this study was conducted from June 2021 to November 2021 due to the COVID restrictions. Thus, only participants with complete vaccinations were enrolled.
Implications to Practice and Recommendations
The results obtained from this study showed promising positive outcomes and benefits of neurobic exercise for community-dwelling older adults with MCI. For further research, it is recommended to examine the sustainable effects of the neurobic exercise intervention among older adults with MCI. Testing the long-term effects of increased memory function on life satisfaction, quality of life, healthy living, fall incidents, and hospitalization or emergency department visits are also recommended. In addition, future studies should investigate the neurobic exercise program in other settings and the impact of neurobic exercise on other cognitions apart from memory. Testing improvement in brain function, such as brain waves and images, is recommended if possible. Also, this neurobic exercise program provides recommendations and practices for older adults residing in the community. Early brain stimulation, such as neurobic exercise, may benefit older persons with early signs of MCI. In addition, involving family members or other members of the community in the game-based brain exercise could also help older adults keep enjoying and doing such activities.
Conclusion
The neurobic exercise program is a new alternative nursing intervention approach, which was found to be effective in enhancing significant memory performance among older adults with MCI who are living in the community. Applying available Thai herbs, aromas, and foods to stimulate the brain was useful. Moreover, the neurobic exercise program is easy for older adults to use to stimulate their brains. It can also help older people make small changes in daily life, transforming routines into mind-building exercises. The neurobic exercise program was found to be effective in reducing or delaying subjective memory decline and improving objective memory in community-dwelling older people with MCI. In addition, neurobic exercise requires no need to find a special time or place to do it because everyday life is a neurobic exercise. The development of the care system for promoting cognitive functions among community-dwelling older persons with MCI should be conducted. Nurses and multidisciplinary teams are recommended to employ the neurobic exercise program alongside other activities to enhance cognitive function among older adults with MCI.