Background
Multiple sclerosis (MS) stands as a prevalent cause of progressive neurological impairment among young adults (McGinley et al., 2021). The global population of people with MS is estimated at 2.8 million (35.9 per 100,000 people). The number of people with MS has risen in all regions of the world, but there are still uncertainties in the prevalence data. The average incidence rate among 75 countries that reported data is 2.1/100,000 people/year, and the average age at diagnosis is 32 years. Women have a twofold higher risk of having MS than men (Walton et al., 2020).
Upper limb deficits, muscle weakness, stiffness, decreased functional performance, and fatigue are among the common clinical symptoms of MS (Agostini et al., 2021; De Sire et al., 2019; Patti et al., 2020; Solaro et al., 2020). Balance disturbance is a noteworthy disability that is frequently seen in MS patients and can increase the risk of falling and limit independence in daily living tasks (Cameron & Nilsagard, 2018; Kraft & Berger, 2021; OMalley et al., 2022; Paolucci et al., 2020). To address these profoundly debilitating challenges, various rehabilitative approaches have been proposed in the existing literature (Amatya et al., 2019; Correale et al., 2021). Technology-based modalities offer promising therapeutic options within the all-encompassing MS treatment framework alongside conventional physiotherapy and rehabilitation interventions (CPRh). Virtual reality (VR) techniques are potentially valuable tools for improving various clinical disorders (Maggio et al., 2022). VR, by simulating an artificial environment akin to the real one, has the potential to enhance interaction and provide multisensory feedback training, thereby augmenting the effectiveness of rehabilitation. Indeed, human balance control is an intricate process involving the integration of multiple sensory inputs, including vestibular, visual, and proprioceptive signals, which are continuously converted into corrective motor actions (Peterka, 2018). Consequently, multisensory augmented reality is promising as a rehabilitation approach for addressing balance impairments in individuals with MS.
Furthermore, VR technology offers a range of features such as head-mounted displays, sound effects, motion tracking devices, and even end-effectors like joysticks or sensors capable of capturing muscle and brain signals (Laver et al., 2015). VR has been integrated into rehabilitative protocols for various neurological diseases, including MS, with promising outcomes (Abou et al., 2020; Liao et al., 2015; Maggio et al., 2022). As a complementary tool in rehabilitation programs, VR allows patients to engage in exergames involving physical exertion while playing video games (Garcia-Bravo et al., 2021; OLoughlin et al., 2020). Exergaming has gained widespread use in rehabilitative programs and clinical studies in recent years (Chesser et al., 2020; Marotta et al., 2022). Exergaming, a form of active video game therapy, can help with dual-task training, boost patient motivation, lessen the monotony of the rehabilitation process, and provide quick feedback. Commercially available exercise games like the Nintendo Wii and Microsoft Kinect have transformed living rooms into pleasurable training spaces during the past ten years (Givon Schaham et al., 2018). The effectiveness of Nintendo Wii Fit in enhancing balance and gait in MS patients has been investigated through clinical and home trials, although the outcomes have produced inconsistent conclusions (Amiri et al., 2018). Researchers have recently begun evaluating exergaming as a rehabilitation tool specifically for individuals with MS. Exergaming has demonstrated acceptability, feasibility, safety, enjoyment, stimulation, and self-motivation as a therapeutic approach (Feys & Straudi, 2019). However, there is limited evidence regarding its efficacy in neurological pathologies (Canning et al., 2020; Costa et al., 2019; Nguyen et al., 2018).
Our study aims to synthesize the available evidence to determine exergaming’s effectiveness in MS, provide a comprehensive understanding of exergaming’s therapeutic potential, and inform clinical practice in this area. This study addresses a nursing problem: how to help MS patients improve their brain and body functions, mood, and quality of life. MS patients face many challenges due to their disorder, and conventional rehabilitation programs may not suit their needs or preferences. This study explores exergaming, a video game exercise, as a novel and relevant nursing intervention for MS patients. Exergaming can be done at home and fit each patient’s needs. Exergaming can improve the outcomes, motivation, adherence, and quality of life of MS patients. This study can advance nursing knowledge and practice in MS care.
Methods
Study Design
This study employed a systematic review and meta-analysis structured following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) (Page et al., 2021) and Cochrane Handbook for Systematic Reviews of Interventions (Higgins et al., 2019).
Literature Search
A comprehensive and systematic exploration was performed across various electronic databases, such as Web of Science, Scopus, PubMed, Cochrane, and EMBASE, from their inception to June 2023. Additionally, an exhaustive examination of references cited within eligible articles and prior meta-analyses relevant to the subject matter was done. The search methodology employed a combined approach utilizing a set of terms including: ((“Multiple sclerosis” OR “sclerosis multiple”) AND (“virtual reality” OR VR OR videogame OR videogames OR exergame* OR “Immersive virtual reality” OR IVR OR “Wii games” OR “Wii game” OR “Nintendo Wii” OR Wii OR “exergaming”)) (See Supplementary File - Table S1).
Eligibility Criteria
The eligibility for inclusion in this systematic review was predicated on satisfying the subsequent criteria: 1) Patients who had confirmed diagnosis of MS; 2) Intervention: Studies used exergaming interventions; 3) Comparison: Studies compared exergaming interventions with either no-intervention or CPRh; 4) Outcomes: Cognitive, Motor, or psychological outcomes; and 5) Study design: studies conducted as randomized controlled trials (RCTs) or observational studies. Articles not published in English, studies involving animals, abstracts lacking complete full-text access, and unpublished data were systematically excluded from consideration in this study.
Data Collection
During the data extraction process, offline data extraction sheets were used to systematically gather relevant details of study characteristics and outcome data from each selected study. The compiled data delineated various aspects, including study arms, study ID, study design, site, age, gender, follow-up duration, disease duration, type of MS, balance and disability scores, inclusion criteria, primary endpoints, and conclusion of each study (see Supplementary File – Table S2). The assessed outcomes were categorized into three main categories: 1) Cognitive outcomes, including Brief Visuospatial Memory Test-Revised (BVMT-R) score, Trail Making Test A and B (TMT B-A) score, Symbol Digit Modalities Test (SDMT) score, Paced Auditory Serial Addition Test (PASAT), and Paced Auditory Serial Addition Test-3 (PASAT-3) score. 2) Motor outcomes: 9-hole peg test score, 6-minute walk test. 10-meter Walk test, Time Up and Go (TUG) test in seconds, and Berg balance scale. 3) Psychological outcomes: Modified fatigue impact scale (MFIS) score and Beck depression inventory.
Risk of Bias Evaluation
The evaluation of retrieved RCTs for interventional studies adhered to the Cochrane Risk of Bias Assessment Tool 1 (ROB1). This tool includes domains such as selection, performance, detection, attrition, reporting, and potential sources of bias. The assessments were stratified into categories of “high,” “low,” or “unclear” risk of bias (Higgins et al., 2019). The quality assessment of the cohort study included in this review was conducted by the National Institutes of Health (NIH) (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools). The assessment process involved assigning scores to the studies, and based on these scores, the quality was categorized. This classification was determined after careful consideration of various factors. In instances of disparity, a third assessor was consulted for resolution.
Data Analysis
Continuous outcomes were consolidated as a mean difference (MD) between the two groups, accompanied by a 95% confidence interval (CI). The fixed-effect model was initially employed in cases where the effect estimate was derived from homogeneous studies. Conversely, if heterogeneity was observed, the random-effect model was utilized. Statistical heterogeneity among studies was assessed using the I2 statistic and chi-squared test. Results indicating chi2-p >0.1 were deemed heterogeneous, while I2 values ≥50% signified substantial heterogeneity (Higgins et al., 2019). Additionally, subgroup analyses were performed whenever possible according to the comparator to exergaming, either with no intervention or CPRh. All statistical analyses were done using Review Manager software (RevMan) version 5.4.
Results
Literature Search Results
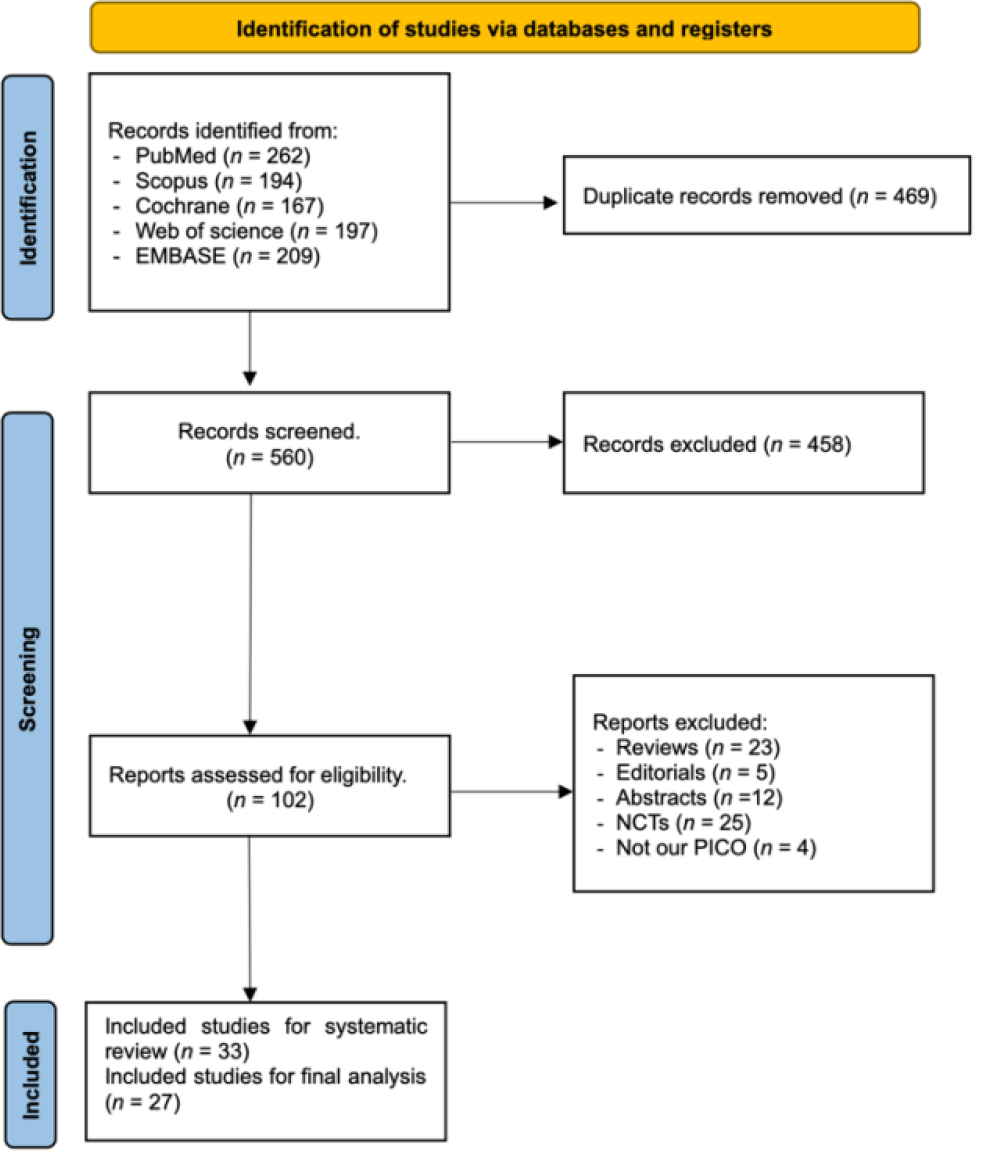
The literature search process across the distinct databases yielded a total of 1029 studies. Subsequently, after eliminating duplicate entries, 560 records were evaluated based on titles and abstracts. Title and abstract screening yields 102 relevant articles for full-text assessment. Based on our performed inclusion criteria, only 33 studies were included in the systematic review (Bove et al., 2021; Brichetto et al., 2013; Calabro et al., 2017; Cuesta-Gómez et al., 2022; Dogan et al., 2023; Eftekharsadat et al., 2015; Galperin et al., 2023; Hoang et al., 2015; Kalron et al., 2016; Khalil et al., 2018; Lamargue et al., 2020; Leonardi et al., 2021; Lozano-Quilis et al., 2014; Maggio et al., 2022; Molhemi et al., 2022; Molhemi et al., 2020; Munari et al., 2020; Nilsagard et al., 2012; Norouzi et al., 2021; Ortiz-Gutiérrez et al., 2013; Ozdogar et al., 2022; Ozdogar et al., 2023; Ozdogar et al., 2020; Ozkul et al., 2020; Pagliari et al., 2021; Peruzzi et al., 2016; Prosperini et al., 2013; Pruszynska et al., 2022; Robinson et al., 2015; Streicher et al., 2018; Tollar et al., 2020; Walino-Paniagua et al., 2019; Yazgan et al., 2020), and 27 were eligible for meta-analysis (Figure 1).
Characteristics of the Included Studies
This study retrieved 32 RCTs and one retrospective cohort consisting of 1421 patients that compared exergaming to no-intervention or CPRh subgroups as interventions for people with MS. The mean age of the participants ranged from 26.39 to 57.4 years, and the percentage of male participants ranged from 0 to 66.7%. The follow-up duration ranged from four to 13 weeks, and the disease duration ranged from 4.83 to 17.7 years. The most common type of MS was relapsing-remitting (RR), followed by secondary progressive (SP) and primary progressive (PP). The balance and disability scores varied depending on the scales used, such as the Berg Balance Scale (BBS), the Expanded Disability Status Scale (EDSS), the Symbol Digit Modalities Test (SDMT), and others. The inclusion criteria also varied depending on the study objectives but generally required a clinical diagnosis of MS in McDonald criteria, an EDSS score between 2 and 6.5, a stable phase of the disease, and written informed consent. The primary endpoints were mostly related to balance, mobility, cognition, fatigue, and quality of life measures. The summary of our included studies can be seen in the supplementary file (Supplementary File - Table S2 Included studies)
Quality Assessment of the Included Studies
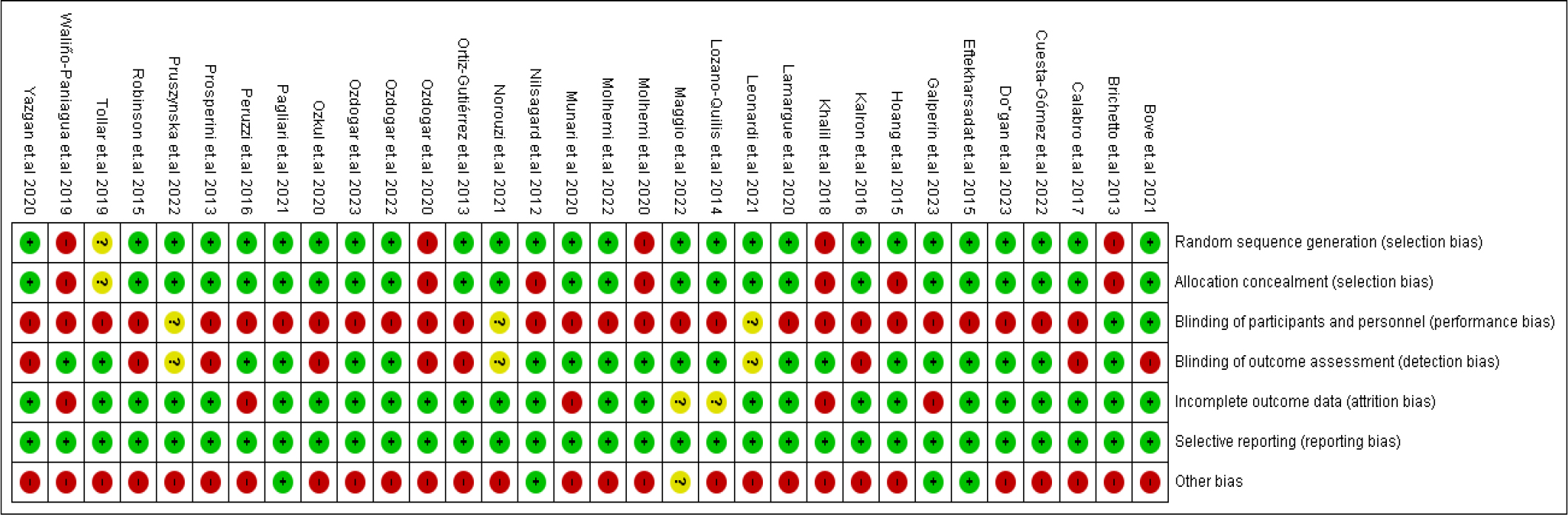
In our included RCTs, the domain with the highest risk of bias was participants’ blinding, while most studies have a low risk of bias regarding selective reporting and incomplete data domains. Streicher et al. (2018) cohort had a fair quality, scoring nine points on the NIH tool for cohort studies. The risk of bias is summarized in Figure 2 and Table 1, and the NIH tool is provided in Table 2.
| Study iD | The Cochrane Collaboration’s tool for assessing risk of bias | |||||||
|---|---|---|---|---|---|---|---|---|
| Low \ High \ Unclear risk of bias | ||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| (Bove et al., 2021) | Low risk | Low risk | Low risk | High risk | Low risk | Low risk | High risk | High risk |
| (Brichetto et al., 2013) | High risk | High risk | Low risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Calabro et al., 2017) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Cuesta-Gómez et al., 2022) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Dogan et al., 2023) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Eftekharsadat et al., 2015) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | Low risk | Low risk |
| (Galperin et al., 2023) | Low risk | Low risk | High risk | Low risk | High risk | Low risk | Low risk | Low risk |
| (Hoang et al., 2015) | Low risk | High risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Kalron et al., 2016) | Low risk | Low risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
| (Khalil et al., 2018) | High risk | High risk | High risk | Low risk | High risk | Low risk | High risk | High risk |
| (Lamargue et al., 2020) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Leonardi et al., 2021) | Low risk | Low risk | Unclear | Unclear | Low risk | Low risk | High risk | High risk |
| (Lozano-Quilis et al., 2014) | Low risk | Low risk | High risk | Low risk | Unclear | Low risk | High risk | High risk |
| (Maggio et al., 2022) | Low risk | Low risk | High risk | Low risk | Unclear | Low risk | Unclear | Unclear |
| (Molhemi et al., 2020) | High risk | High risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Molhemi et al., 2022) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Munari et al., 2020) | Low risk | Low risk | High risk | Low risk | High risk | Low risk | High risk | High risk |
| (Nilsagard et al., 2012) | Low risk | High risk | High risk | Low risk | Low risk | Low risk | Low risk | High risk |
| (Norouzi et al., 2021) | Low risk | Low risk | Unclear | Unclear | Low risk | Low risk | High risk | High risk |
| (Ortiz-Gutiérrez et al., 2013) | Low risk | Low risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
| (Ozdogar et al., 2020) | High risk | High risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
| (Ozdogar et al., 2022) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Ozdogar et al., 2023) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Ozkul et al., 2020) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | High risk | High risk |
| (Pagliari et al., 2021) | Low risk | Low risk | High risk | Low risk | Low risk | Low risk | Low risk | Low risk |
| (Peruzzi et al., 2016) | Low risk | Low risk | High risk | Low risk | High risk | Low risk | High risk | High risk |
| (Prosperini et al., 2013) | Low risk | Low risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
| (Pruszynska et al., 2022) | Low risk | Low risk | Unclear | Unclear | Low risk | Low risk | High risk | High risk |
| (Robinson et al., 2015) | Low risk | Low risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
| (Tollar et al., 2020) | Unclear | Unclear | High risk | Low risk | Low risk | Low risk | High risk | Unclear |
| (Walino-Paniagua et al., 2019) | High risk | High risk | High risk | Low risk | High risk | Low risk | High risk | High risk |
| (Yazgan et al., 2020) | Low risk | Low risk | High risk | High risk | Low risk | Low risk | High risk | High risk |
Note: 1 Random sequence generation (selection bias) | 2 Allocation concealment (selection bias) | 3 Blinding of participants and personnel (performance bias) | 4 Blinding of outcome assessment (Detection bias) | 5 Incomplete outcome data (attrition bias) | 6 Selective reporting (reporting bias) | 7 Other Bias | 8 Author judgment
| Study iD | NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies | Rating | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Yes / No / Not reported (NR) or cannot determine (CD) or not applicable (NA) | ||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | Total Score | ||
| (Streicher et al., 2018) | Yes | Yes | Yes | NR | NR | Yes | Yes | NR | Yes | NR | Yes | NR | Yes | Yes | 9 | Fair |
Note: Quality rating: good (11-14), fair (7.5-10.5), poor (0-7) | Yes = 1, No = 0.5, NR, NA, & CD = 0
1. Was the research question or objective in this paper clearly stated?
2. Were eligibility/selection criteria for the study population prespecified and clearly described?
3. Were the participants in the study representative of those who would be eligible for the test/service/intervention in the general or clinical population of interest?
4. Were all eligible participants that met the prespecified entry criteria enrolled?
5. Was the sample size sufficiently large to provide confidence in the findings?
6. For the analyses in this paper, were the exposure(s) of interest measured prior to the outcome(s) being measured?
7. Was the time frame sufficient so that one could reasonably expect to see an association between exposure and outcome if it existed?
8. For exposures that can vary in amount or level, did the study examine different levels of exposure as related to the outcome (e.g., categories of exposure or exposure measured as continuous variable)?
9. Were the exposure measures (independent variables) clearly defined, valid, reliable, and implemented consistently across all study participants?
10. Was the exposure(s) assessed more than once over time?
11. Were the outcome measures prespecified, clearly defined, valid, reliable, and assessed consistently across all study participants?
12. Were the people assessing the outcomes blinded to the participants’ exposures/interventions?
13. Was the loss to follow-up after baseline 20% or less? Were those lost to follow-up accounted for in the analysis?
14. Were key potential confounding variables measured and adjusted statistically for their impact on the relationship between exposure(s) and outcome(s)?
Outcomes
Motor outcomes
Regarding walking tests, the 6-min walking test showed significantly better results in the exergaming group than the no-intervention group with MD and 95% CI = (25.53 [6.87, 44.19], p = 0.007), as shown in Figure 3.
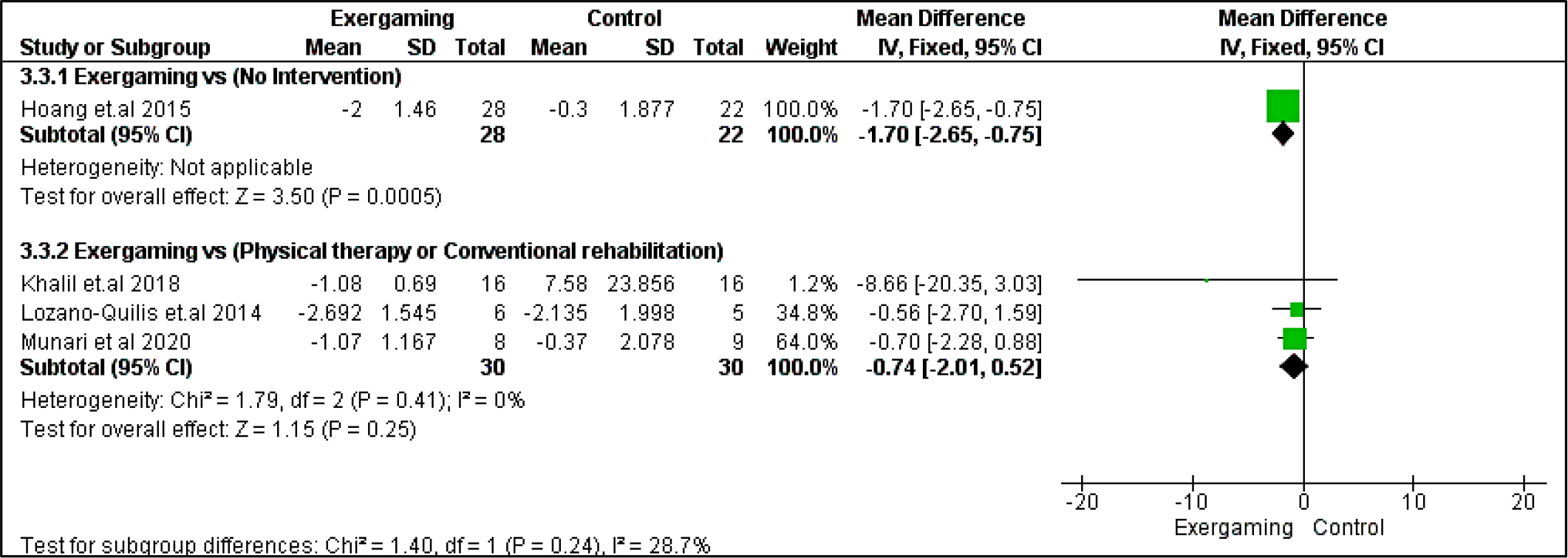
In the 10 m walking test, we did not detect any significant difference between the compared interventions in the CPRh subgroup (MD = -0.74 [-2.01, 0.52], p = 0.25). Regarding heterogeneity, the CPRh subgroup was homogenous with I2 and Chi2-p (0%, 0.41) (Figure 4).
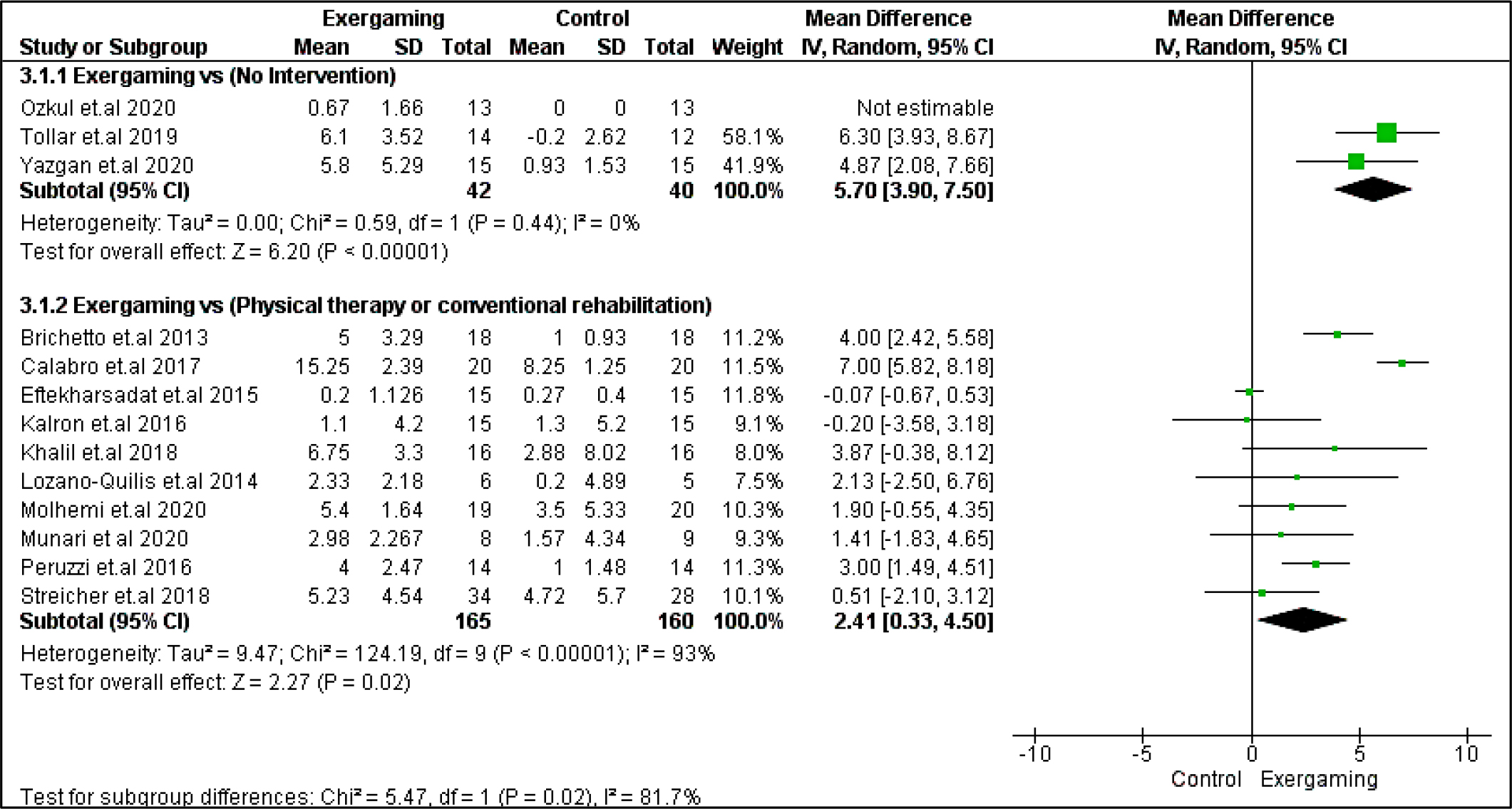
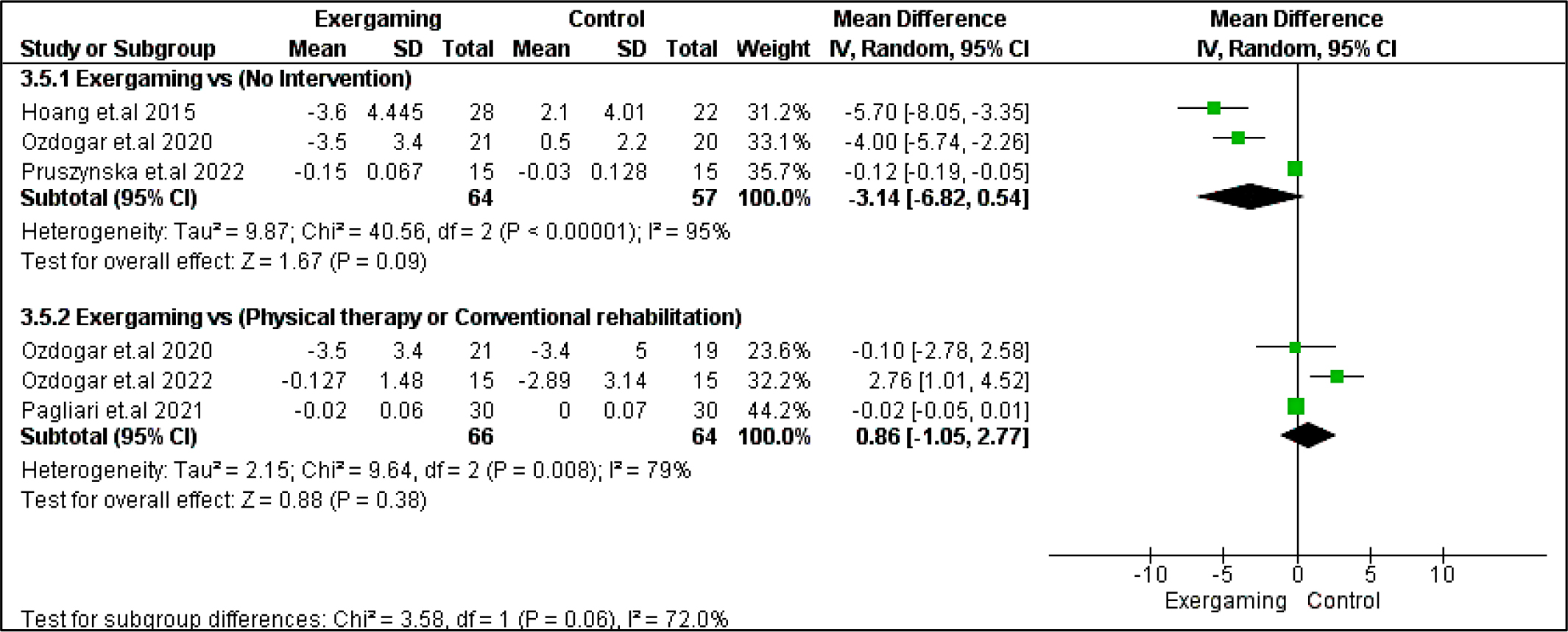
Berg balance scale score in both studied subgroups and TUG in no-intervention subgroups significantly (p > 0.05) favored the exergaming intervention group with MD, and their relevant 95% CI were as follows: 5.70 [3.90, 7.50], 2.41 [0.33, 4.50], -1.20 [-1.60, -0.81], respectively. Regarding the 9-hole peg test, the results were indifferent between interventions in the two studied subgroups. Pooled MD for no-intervention subgroups was (3.14 [-6.82, 0.54], p = 0.09), and for the CPRh subgroup was (0.86 [-1.05, 2.77], p = 0.38). Forest plots for these motor outcomes are shown in Figure 5, Figure 6, and Figure 7, respectively.
Regarding heterogeneity, the CPRh subgroup was homogenous in TUG with I2 and Chi2-p = (30%, 0.19). In the no-intervention subgroup, Berg balance and TUG studies were homogenous with I2 and Chi2-p = (0%, 0.44) and (36%, 0.18), respectively. However, the rest of the outcomes were heterogeneous (Chi2-p <0.1), as shown in Figure 5, Figure 6, and Figure 7.
Cognitive outcomes
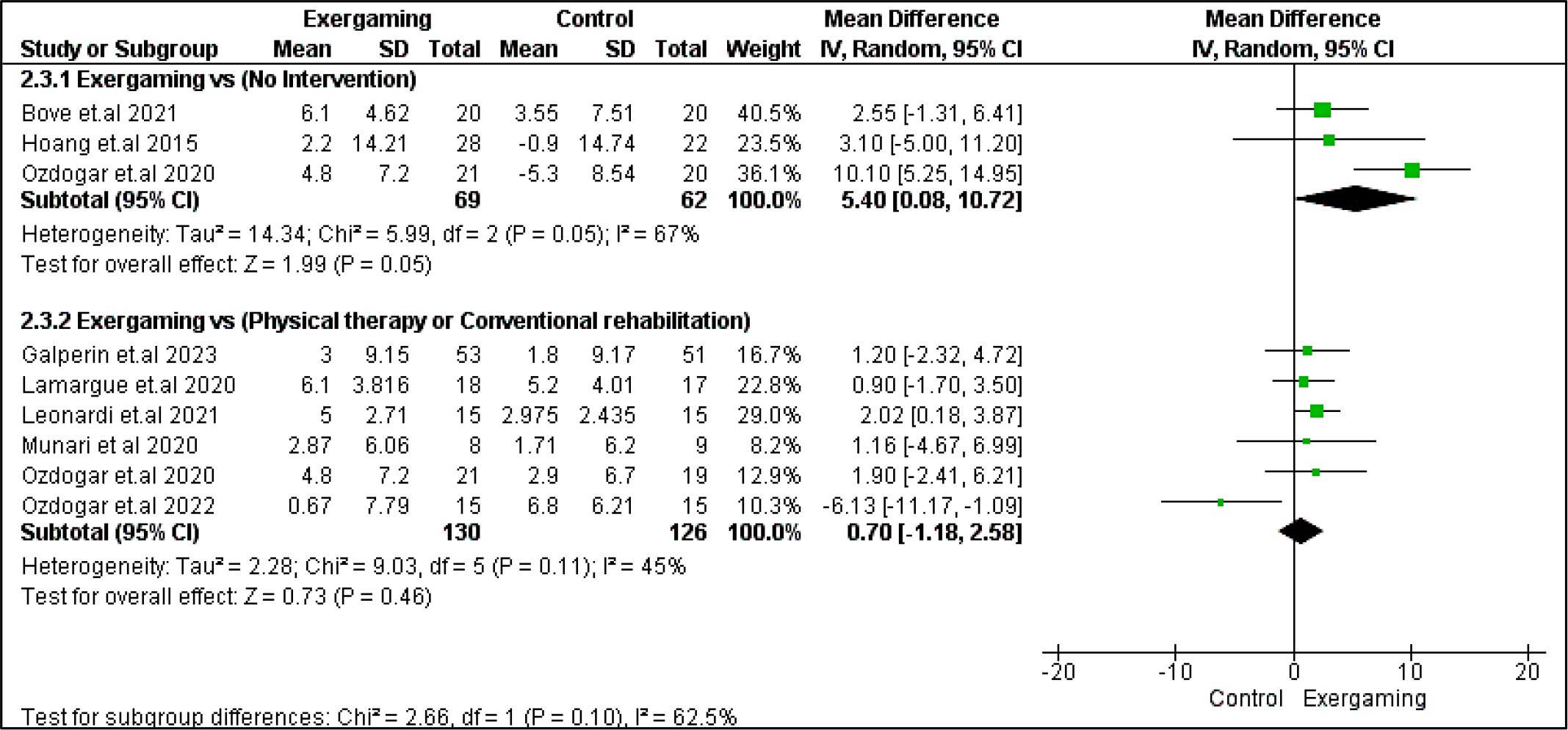
SDMT was the only outcome among cognitive outcomes to show a statistically significant difference in the no-intervention subgroup with MD = 5.40 [0.08, 10.72] and p = 0.05, favoring exergaming. However, no significant difference was revealed when comparing CPRh to exergaming (MD = 0.70 [-1.18, 2.58], p = 0.46). The pooled studies in the subgroup of no-intervention were heterogeneous with I2 and Chi2-p as follows: (67% and 0.05) (Figure 8).
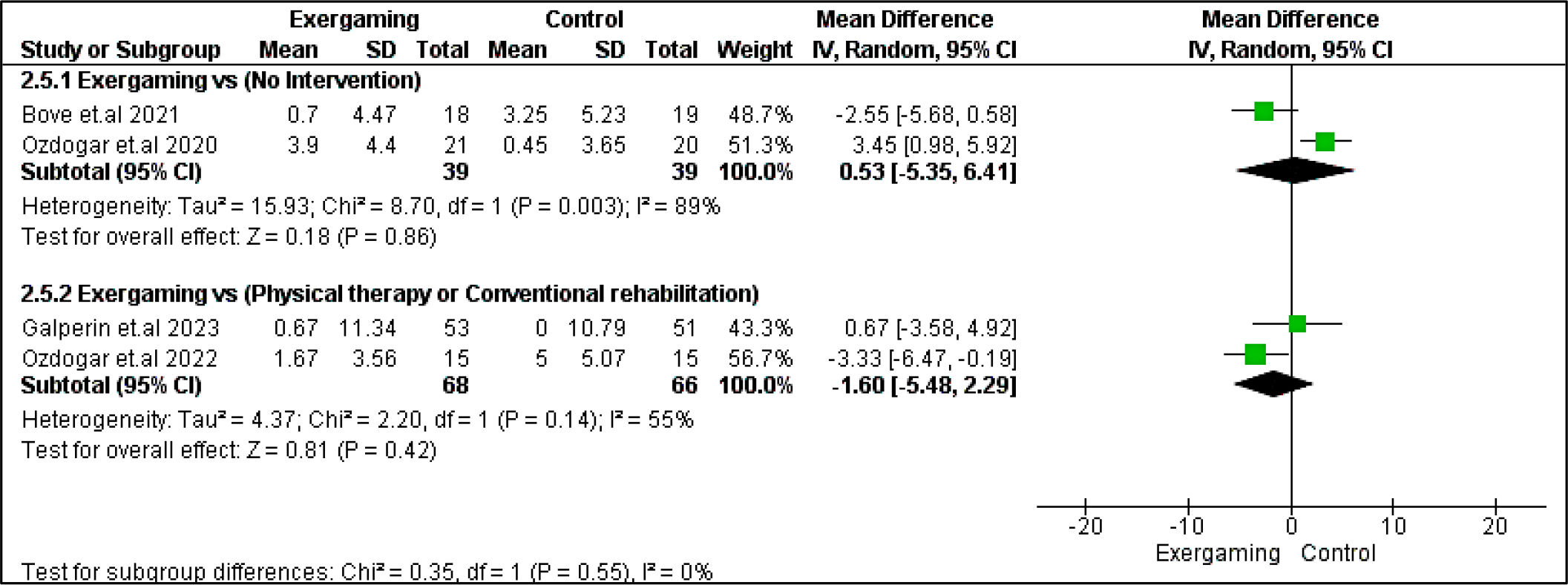
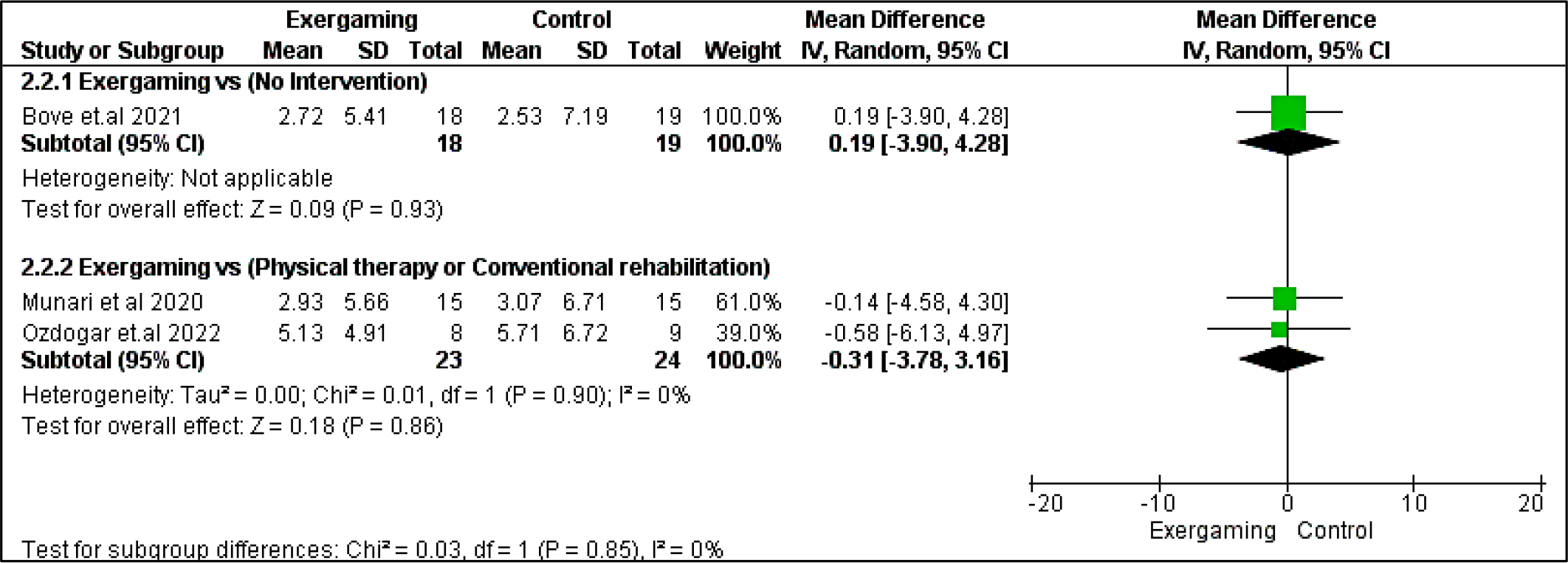
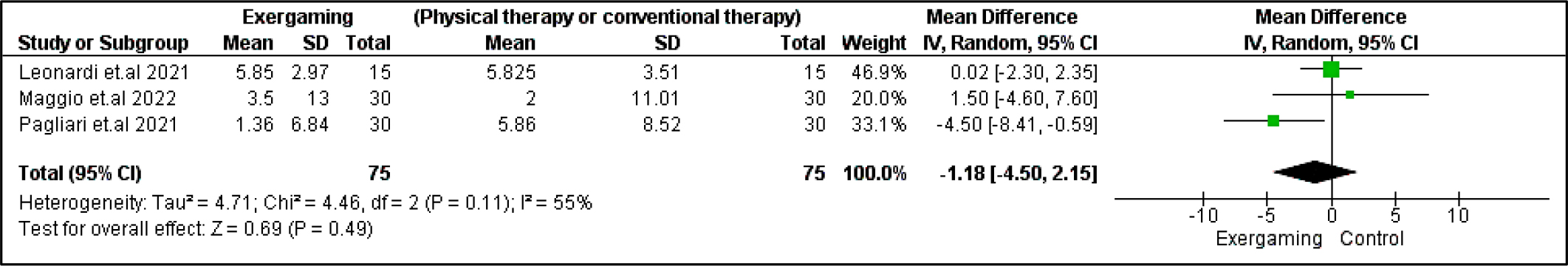
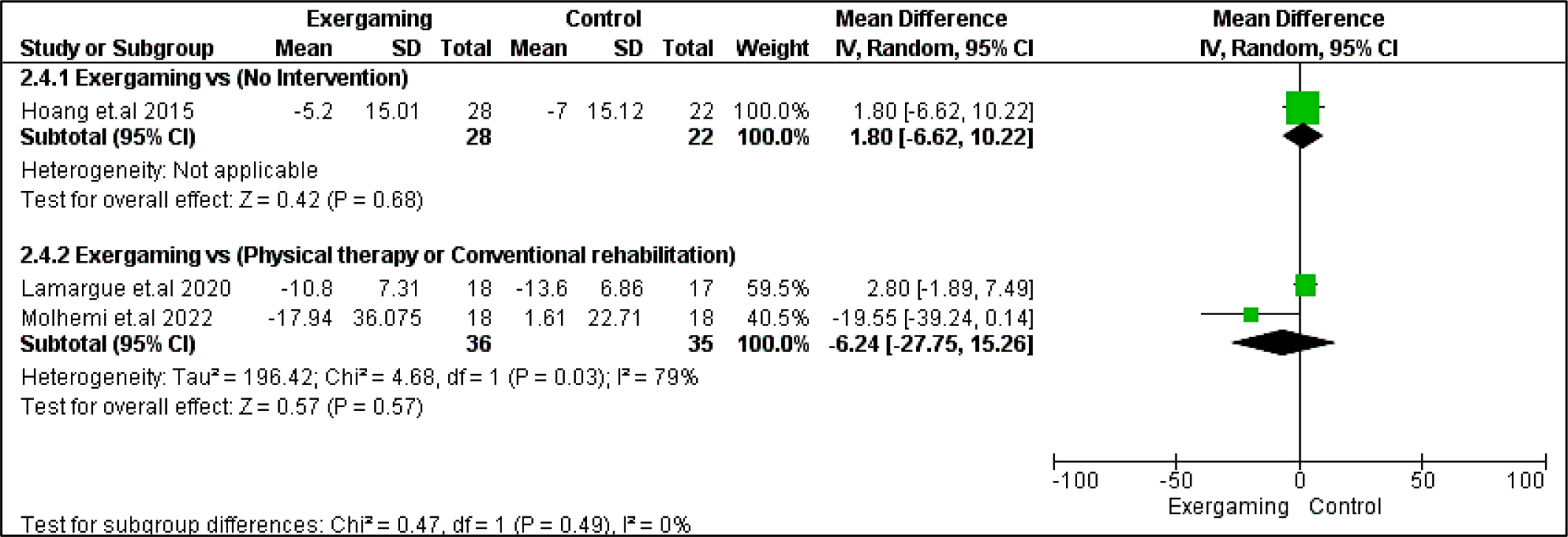
Regarding BVMT-R outcome, no significant difference was detected between exergaming and no-intervention nor CPRh subgroups with MD and 95% CI = (0.53 [-5.35, 6.41], p = 0.86) and (-1.60 [-5.48, 2.29], p =0.42), respectively. Also, PASAT and PASAT-3 pooled analyses did not reveal any remarking difference between exergaming and CPRh, MD = (-0.31 [-3.78, 3.16], p = 0.86) and (-1.18 [-4.50, 2.15], p = 0.49), respectively. As to the TMT B-A score, the difference between the two compared groups was insignificant; MD in the physical and rehabilitation subgroup was ( -6.24 [-27.75, 15.26], p = 0.57). Forest plots for cognitive outcomes are shown in Figure 9, Figure 10, Figure 11, and Figure 12, respectively.
Regarding heterogeneity between studies assessing cognitive outcomes, the pooled studies in the subgroup of no-intervention in BVMT-R were heterogeneous with I2 and Chi2-p as follows: (89% and 0.003). Meanwhile, in the CPRh subgroup, pooled studies assessing TMT B-A showed heterogeneity with I2 and Chi2-p = (79% and 0.03). However, the rest of the outcomes were homogeneous (Chi2-p <0.1).
Psychological outcomes
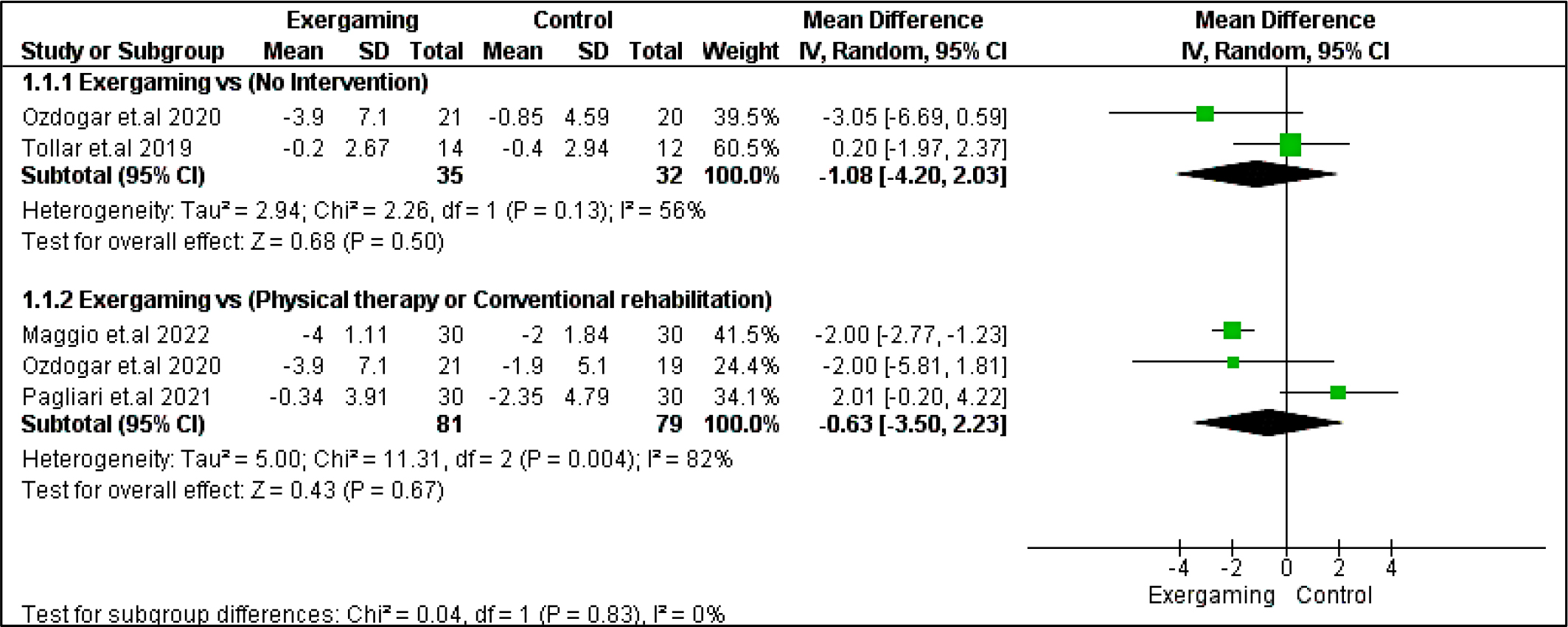
Regarding Beck depression inventory outcome, no significant difference existed between exergaming and either no-intervention or CPRh subgroup with MD = -1.08 [-4.20, 2.03], p = 0.50, and -0.63 [-3.50, 2.23], p = 0.67, respectively. The pooled studies for the no-intervention subgroup were homogenous with Chi2- p >0.10, While the pooled studies in the CPRh subgroup were heterogenous, and the detected heterogeneity could not be solved (Figure 13).
On the other hand, the MFIS score outcome favored exergaming in the CPRh subgroup = -9.66 [-16.26, -3.07], p = 0.004. Meanwhile, there was no significant statistical difference in the no-intervention subgroup, with MD in the no-intervention subgroup = -5.04 [-10.18, 0.11], p = 0.06, and the pooled studies for the no-intervention subgroup were homogenous with Chi2-p< 0.10. Meanwhile, the pooled studies in the CPRh subgroup were heterogeneous, and the detected heterogeneity couldn’t be solved (Figure 14).
Discussion
The findings revealed no significant differences in cognitive outcomes between the exergaming and the no-intervention group or the CPRh subgroups. However, the SDMT showed a statistically significant difference in favor of the exergaming in the no-intervention subgroup. In motor outcomes, the exergaming demonstrated better results in the 6-minute walking test compared to the no-intervention group. In contrast, the 10-meter walking test showed no significant difference between the interventions in the CPRh subgroup. The Berg Balance Scale score and TUG test favored the exergaming in both subgroups. The 9-hole peg test did not show a significant difference between the interventions. In terms of psychological outcomes, the Beck Depression Inventory did not reveal any significant differences, while the MFIS favored the exergaming in the CPRh subgroup.
Most of our previous findings align with the outcomes reported in the existing literature. Benedict et al. (2021) elucidated that the SDMT score experiences a decline during relapse, followed by a subsequent return to baseline levels after three months. Conversely, Giedraitiene et al. (2018) postulated that the enhancement of cognitive functions reaches its zenith within the initial month after relapse treatment. Additionally, in their investigation encompassing 36 individuals diagnosed with relapsing multiple sclerosis during the relapse phase and 17 individuals in the remission phase, Ozakbas et al. (2004) utilized the PASAT to evaluate cognitive function. Their findings revealed a noteworthy improvement on day five, whereas no significant alteration was observed on day 30. The discrepancies observed among these studies may arise from the dearth of sensitive and standardized assessment methodologies for evaluating cognitive functions during relapse. Moreover, the absence of baseline assessments in individuals with MS obscures the accurate detection of cognitive impairment during the relapse period.
Balance impairment is a highly debilitating symptom in individuals with MS (Maggio et al., 2022; Pearson et al., 2015). Physiotherapy programs have been reported to have a modest yet significant impact on the balance among individuals with MS who have mild-to-moderate levels of disability (Motl et al., 2017; Paltamaa et al., 2012). With the increasing utilization of technology in rehabilitation, exergaming has emerged as a promising approach within virtual reality-based neurologic rehabilitation. Integrating virtual reality tools in rehabilitation can enhance motor functions, particularly balance and gait, as well as cognitive abilities. Furthermore, it can boost self-efficacy, motivation, and participation in individuals with MS (Maggio et al., 2019).
According to Yazgan et al. (2020), both intervention groups who did exergaming significantly improved their balance parameters compared to the control group. Also, in line with our findings, Brichetto et al. (2013) reported noteworthy enhancements in the Berg Balance Scale. Prosperini et al. (2013) proposed that home-based Nintendo Wii training, characterized by its affordability and user-friendly interface providing diverse exercise options, could potentially serve as an effective and engaging approach for balance rehabilitation among individuals with MS. Nevertheless, prudent measures should be taken to mitigate the risk of injuries associated with training sessions. On the contrary, Nilsagard et al. (2007) observed no statistically significant variance in balance parameters between the groups utilizing Nintendo Wii Fit and the control group. The inconsistencies observed among these studies are likely attributable to methodological differences, specifically variations in session duration, frequency, and the utilization of different assessment scales.
The level of functionality holds significant importance for individuals with MS as it enables them to perform daily living activities independently and facilitates their participation in various domains. Exercise has improved walking speed and endurance in MS (Pearson et al., 2015). Yazgan et al. (2020) observed functional improvement in the intervention groups following treatment with Nintendo Wii Fit. These findings were also observed in previous research utilizing Nintendo Wii Fit in MS, which demonstrated improvements in balance and walking performance (Brichetto et al., 2013; Plow & Finlayson, 2011; Prosperini et al., 2013). Likewise, investigations involving the Balance Trainer across diverse patient cohorts have documented statistically notable improvements in parameters such as the TUG and 10-meter walk tests (Goljar et al., 2010; Matjac✓ ić & Zupan, 2006). The enhancements in functional mobility among the intervention groups can be attributed to multifaceted factors, including augmented confidence in mobility and the physiological impacts stemming from the training regimen (Brichetto et al., 2013; Plow & Finlayson, 2011). Furthermore, research indicates that these variables are subject to influence by the psychological impacts associated with exergaming (Lee et al., 2017; Song et al., 2013).
Implications
The findings of this meta-analysis have several implications for the clinical practice, particularly nursing practice, of treating MS patients. First, exergaming can be a feasible and effective alternative to conventional rehabilitation programs for improving cognitive and motor outcomes in MS patients, especially during the relapse phase. Exergaming can provide a stimulating and engaging environment that can enhance the motivation and adherence of MS patients to their rehabilitation regimen. Moreover, exergaming can be easily implemented at home, reducing travel and cost burdens for MS patients and their caregivers. Second, exergaming can be tailored to the individual needs and preferences of MS patients, as well as their level of disability and fatigue. Exergaming can offer a variety of exercises that can target different cognitive and motor domains, such as attention, memory, processing speed, balance, gait, and coordination. Exergaming can also adjust the difficulty and feedback of the exercises according to the performance and progress of MS patients. Third, exergaming can have a positive impact on the psychological well-being of MS patients, as it can reduce the symptoms of depression and fatigue and improve the quality of life and self-efficacy. Exergaming can foster a sense of achievement and enjoyment for MS patients and a sense of social connectedness and support if they play with others.
Therefore, exergaming can be a valuable addition to the current treatment options for MS patients, as it can offer a comprehensive and personalized approach to address the multifaceted challenges of this chronic and progressive neurological disorder. However, further research is needed to establish the optimal parameters and protocols of exergaming for MS patients, such as the frequency, intensity, duration, and type of exercises, as well as the long-term effects and safety of exergaming. Additionally, more studies are needed to compare the efficacy of exergaming with other forms of rehabilitation, such as physical therapy, occupational therapy, cognitive training, and pharmacological interventions.
Strengths and Limitations
To our knowledge, our study is the most exhaustive meta-analysis conducted thus far concerning the impact of exergaming on patients with MS. The outcomes derived from existing studies were meticulously scrutinized and comprehensively deliberated upon in our analysis. Our study boasts a significantly larger sample size than previous meta-analyses (Calafiore et al., 2021; Cortes-Perez et al., 2023; Perrochon et al., 2019). This increased sample size confers enhanced statistical power to our findings, reducing the likelihood of type II errors (false negative results) and bolstering the credibility of our evidence. However, our study was not free of limitations. First, we had a lot of confounders contributing to heterogeneity presented in our study; however, the available data did not help us to stratify the type of exergaming or the type of rehabilitation or physical therapy, type of MS, and according to the severity of the disease. Most of the studies we included were not of high quality and had major drawbacks, such as small sample sizes, and many of them were pilot trials. Therefore, we need to conduct high-quality trials on that topic, considering mentioned confounders that could bias the results.
Conclusion
Our meta-analysis suggests that exergaming may contribute significantly to improved cognitive, motor, and psychological outcomes in patients with MS. Exergaming demonstrated superior performance in the SDMT, 6-minute walking test, Berg Balance Scale score, TUG test, and the MFIS. However, the cognitive and certain motor outcomes, such as the 10-meter walking and 9-hole peg tests, showed no significant difference with conventional rehabilitation methods. Notwithstanding the limitations and heterogeneity of the studies, these findings underscore the potential of exergaming as a beneficial supplement to traditional rehabilitation approaches for MS patients, necessitating further high-quality trials to substantiate these results.